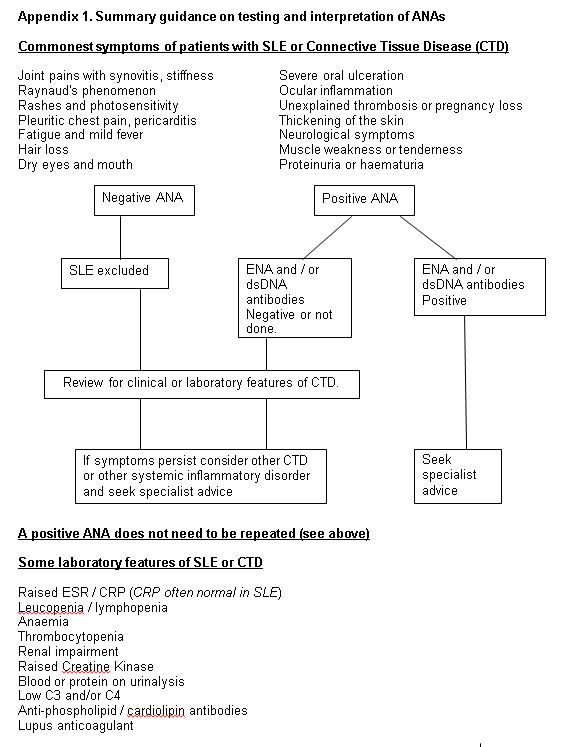
Pathology Guidelines Antinuclear antibody (ANA) testing




- Created on .
- Last updated on .
Written by Stephen Rimmer on .





Written by Stephen Rimmer on .
The QuantiFERON-TB Gold In Tube and the T-SPOT are two in-vitro tests for measuring cell-mediated immune responses to peptide antigens from mycobacteria. These antigens, ESAT-6, CFP-10 and TB7.7 (p4) (which is used only in QFT-G) are absent from all BCG strains and from most non tubercular mycobacterial strains (NTMs) with the exception of M. kansasii, M. szulgai and M. marinum. Individuals infected with M. tuberculosis complex organisms (M. tuberculosis, M. bovis, M. africanum, M. microti, M. canetti) have mononuclear cells in their blood that recognise these mycobacterial antigens. This recognition process leads to the stimulation and secretion of IFN-γfrom sensitized T-cells. The detection and quantification of IFN-γ, measured by enzyme-linked immunoassay (Quantiferon) or enzyme-linked immunospot (T-SPOT), forms the basis of these tests – detectable as early as two weeks after infection with M. tuberculosis.
Both tests are intended for use in conjunction with risk assessment, radiography, and other medical and diagnostic evaluations. QuantiFERON Gold-IT is the laboratory recommended method for routine screening of patients with a normal lymphocyte count. T spots are recommended in specific circumstances (see below).
Read more: INTERFERON GAMMA RELEASE ASSAYS (IGRA) FOR LATENT TB TESTING.
Written by Craig Webster on .
The basic approach to clinical problems is suggested.
The medical staff of the department are happy to advise with the investigation of any problems.
Read more: Immunology Test Profiles for Common Clinical Problems
Written by Stephen Rimmer on .
This Algorithm describes the actions to be taken on the discovery of a new monoclonal protein in a patient.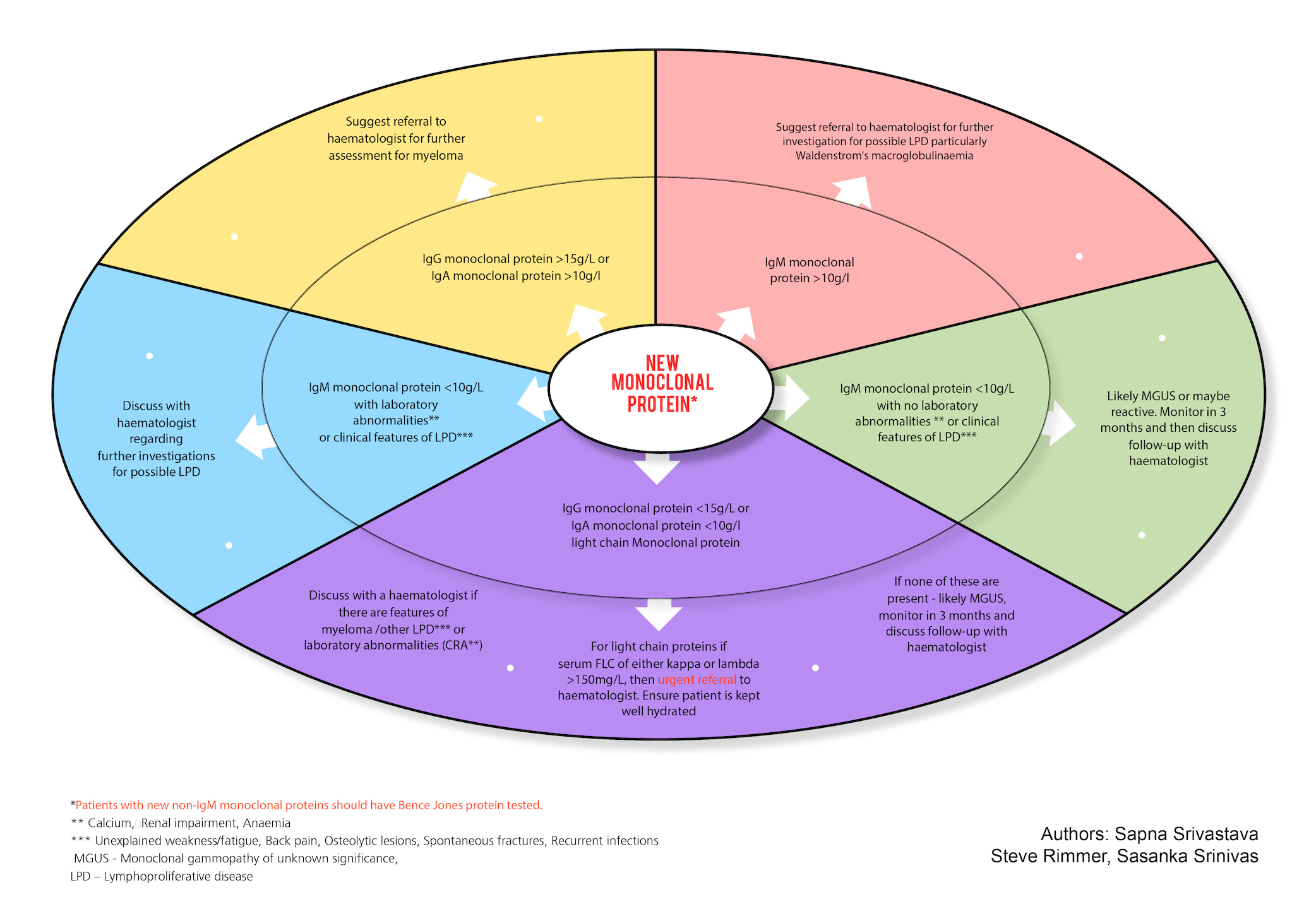
Written by Craig Webster on .
Measurement of IgE to specified allergens can help to confirm allergies suspected on clinical grounds. Results must be interpreted in the context of a detailed clinical history, and neither positive or negative results are diagnostic in isolation. The allergens to be tested must be specified on the request. This test is not suitable for broad "allergy screening" in the absence of a history suggestive of an allergic reaction.
In many cases skin prick testing provides a safe and simple alternative to specific IgE measurement, but this does require referral.
Referrals are accepted for the investigation and management of patients who have suffered anaphylaxis and patients with the the following allergic conditions:
Clear allergic precipitants can be identified in only a small proportion of cases. Patients may respond well to regular treatment with antihistamines.
Skin-prick testing for a range of food allergens is available. "Screening" is not appropriate, and diet and symptom diaries often help to identify suspected foods for testing. Patients with life-threatening reactions benefit from detailed advice and individualised management plans.
Skin prick testing can help to identify significant aeroallergens in these patients, which may held in planning medical management. Pollen desensitisation may be offered under certain circumstances: patients will only be considered if adequate medical therapy has failed, and certain other suitability criteria are fulfilled.
Assessment of patients who have suffered severe reactions to bee or wasp stings is offered. In some cases desensitisation immunotherapy is indicated to prevent anaphylaxis in individuals at particular risk.
Suspected antibiotic allergy (especially to penicillin) is common, and can usually be circumvented by the choice of an alternative antibiotic. In exceptional circumstances allergy testing may be appropriate. It is often important to exclude or identify allergy to local and general anaesthetic agents, so that future routine or emergency treatment can be given safely. A detailed history of the suspected anaesthetic reaction and the anaesthetic agents used are vital to successful assessment. Testing of serum tryptase levels immediately after an anaphylactic episode often aids diagnosis (see Laboratory Services).